Research Articles
Anchorage-Dependent vs. Suspension Cell Culture: A Complete Guide for Bioprocessing
This comprehensive guide explores the critical distinctions between anchorage-dependent (adherent) and suspension cell culture systems, tailored for researchers and bioprocessing professionals.
A Practical Guide to MIQE Guidelines for Diagnostic qPCR: Ensuring Reliable and Reproducible Assay Validation
This comprehensive guide details the application of the Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE) guidelines specifically for diagnostic qPCR assay validation.
The Complete Guide to MIQE Guidelines: Designing and Validating Robust qPCR Assays for Clinical and Biomedical Research
This comprehensive guide details the application of the Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE) guidelines for assay design and validation.
MIQE Guidelines in Clinical Chemistry: Implementing Rigorous Standards for Reproducible Biomarker Research and Diagnostic Assay Development
This article provides a comprehensive guide to the Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE) guidelines, explicitly tailored for clinical chemistry applications.
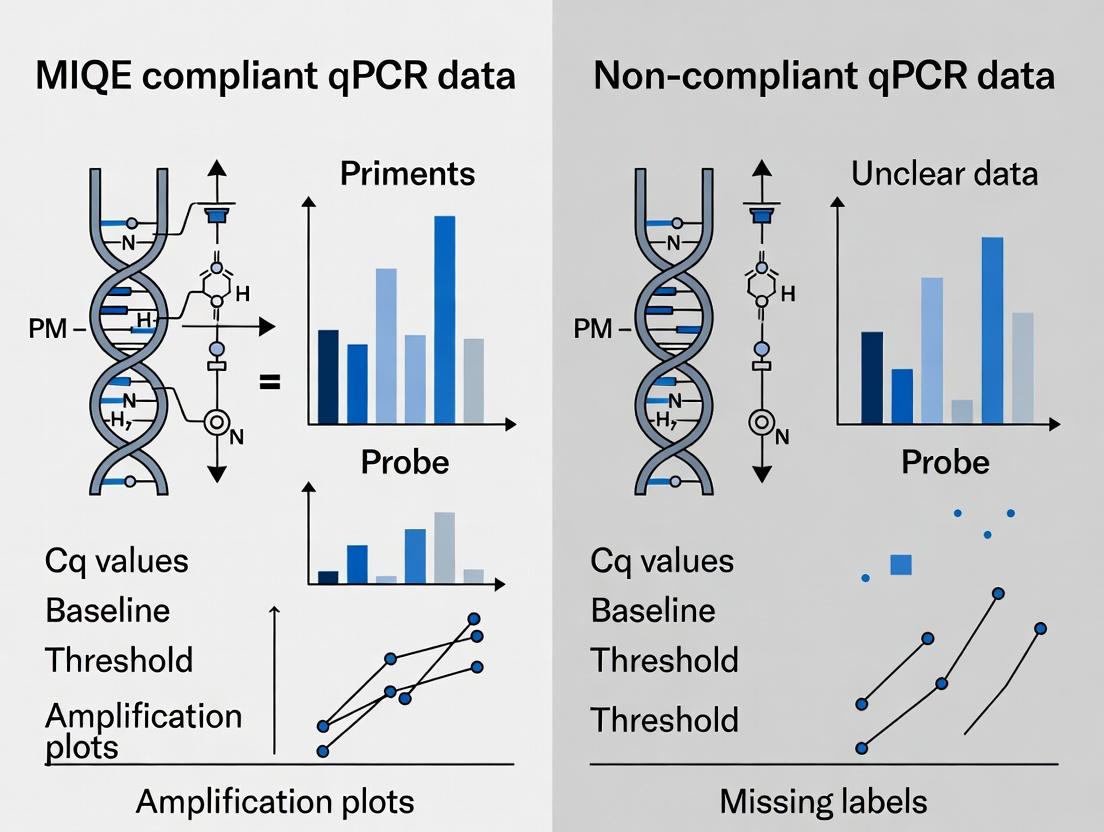
MIQE Guidelines in qPCR: The Critical Difference Between Publishable and Questionable Data
This article explores the profound impact of MIQE (Minimum Information for Publication of Quantitative Real-Time PCR Experiments) compliance on data integrity in biomedical research.
MIQE-Compliant Primer-Probe Sequences: Essential Disclosure for Reproducible qPCR in Biomedical Research
This article provides a comprehensive guide to MIQE-compliant primer and probe sequence disclosure for quantitative PCR (qPCR) and digital PCR (dPCR).
MIQE-Compliant RNA QC for qPCR: The Complete Guide to Accurate Gene Expression Analysis
Accurate quantitative PCR (qPCR) begins with high-quality RNA.
MIQE 2.0 2025 Update: The Essential Guide to Reproducible qPCR for Biomedical Research
This article provides a comprehensive summary of the 2025 update to the MIQE (Minimum Information for Publication of Quantitative Real-Time PCR Experiments) guidelines.
Validating MALDI-TOF MS for Clinical Diagnostics: A Complete Guide for Microbiology Laboratory Implementation
This comprehensive article explores the critical validation process for implementing Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS) in clinical microbiology laboratories.
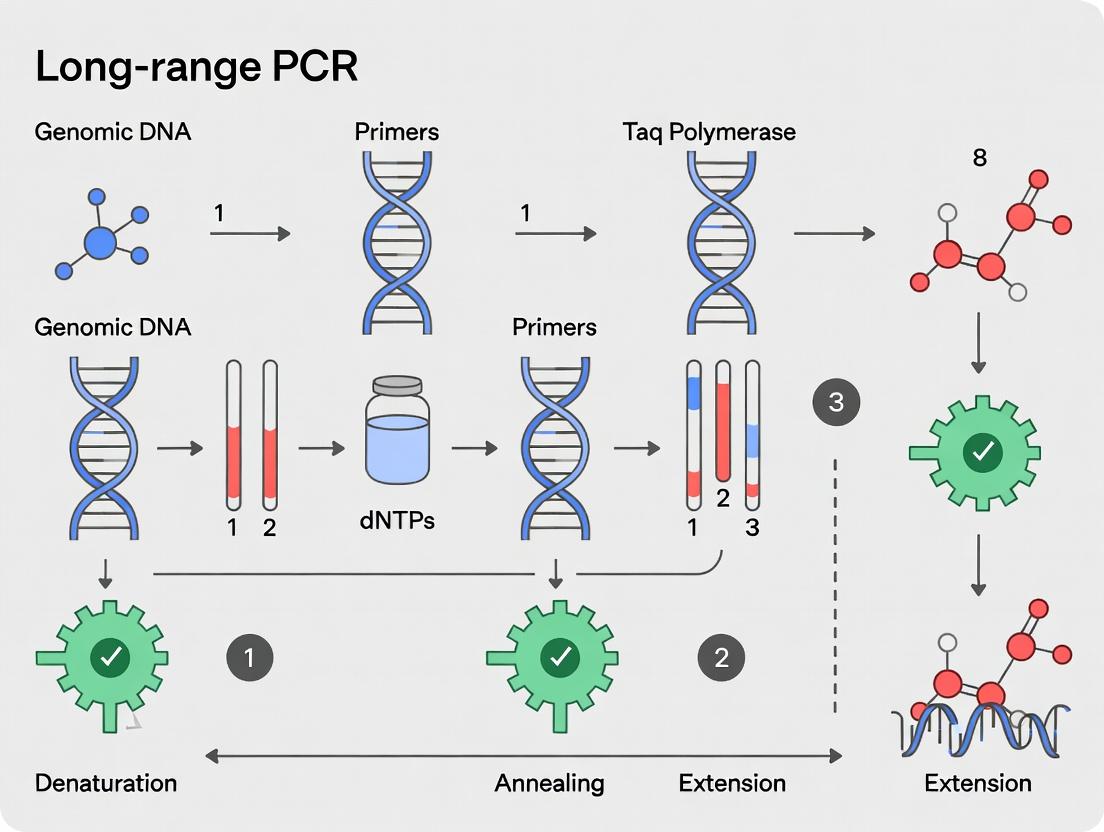
Mastering Long-Range PCR: A Comprehensive Protocol for High-Fidelity Genomic DNA Amplification in Research
This detailed guide provides researchers, scientists, and drug development professionals with a complete framework for successful long-range PCR amplification of genomic DNA.