Research Articles
Method Verification in Microbiology Labs: When and How to Ensure Regulatory Compliance
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on the critical requirements for method verification in microbiology laboratories.
Fundamentals of Microbiological Method Verification: A Comprehensive Guide to Study Design for Reliable Lab Results
This article provides researchers, scientists, and drug development professionals with a comprehensive framework for designing robust microbiological method verification studies.
Fitness for Purpose in Microbiological Methods: A Comprehensive Guide for Robust and Reliable Testing
This article provides researchers, scientists, and drug development professionals with a comprehensive understanding of 'fitness for purpose' for microbiological methods.
Navigating FDA Microbiology Test Verification: A Guide to Compliance and Best Practices for 2025
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on meeting U.S.
A Comprehensive Guide to Designing a Robust Microbiological Verification Study for Pharmaceutical and Biomedical Research
This article provides a comprehensive, step-by-step framework for researchers, scientists, and drug development professionals to design and execute a robust microbiological verification study.
Navigating ISO 15189:2022: A Practical Guide to Microbiology Test Verification and Validation
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on implementing the updated ISO 15189:2022 requirements for verification and validation in clinical microbiology laboratories.
Method Verification in Clinical Microbiology: A Comprehensive Guide for Laboratory Professionals
This article provides a definitive guide to method verification in clinical microbiology, a mandatory process for implementing unmodified FDA-cleared tests.
Mastering CLSI EP12: A Comprehensive Guide to Evaluating Qualitative Binary Test Performance
This guide provides researchers, scientists, and drug development professionals with a complete framework for evaluating qualitative, binary output examinations based on the CLSI EP12 protocol.
ISO 16140 Method Verification: The Complete Guide for Pharmaceutical and Clinical Research Professionals
This guide provides researchers, scientists, and drug development professionals with a comprehensive understanding of the ISO 16140 series for microbiological method validation and verification.
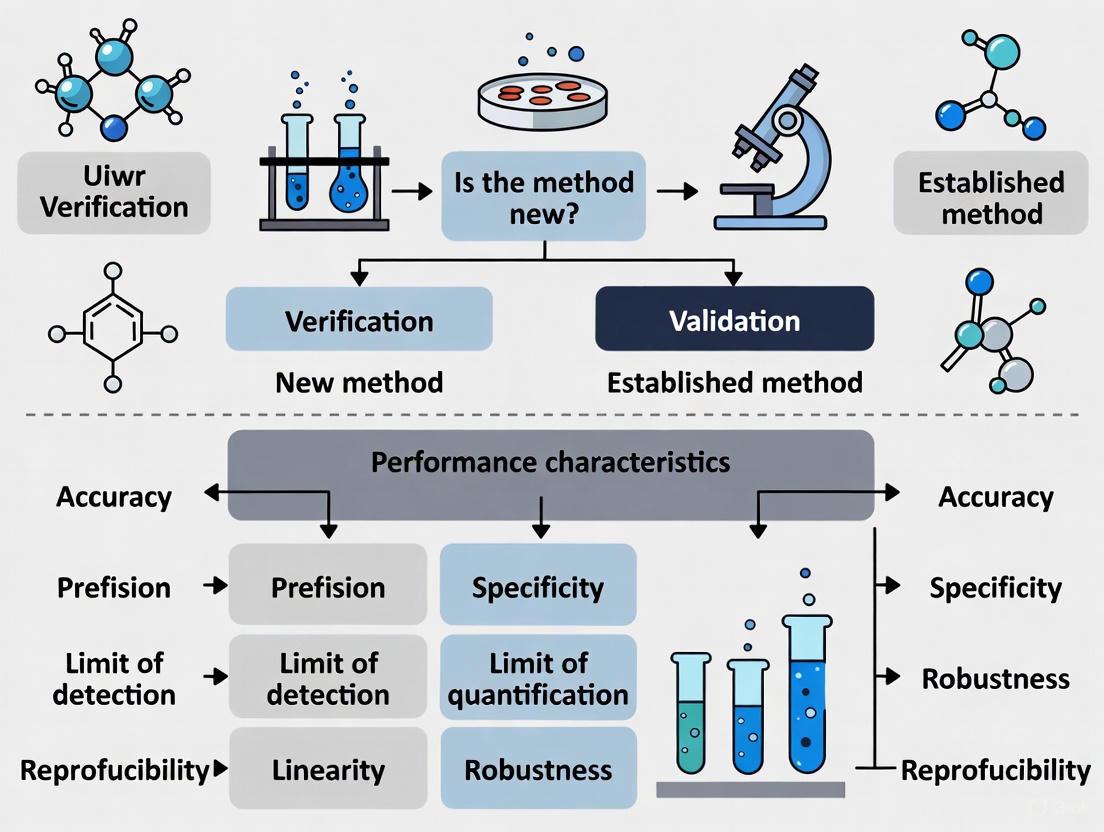
Microbiological Method Verification vs Validation: A Strategic Guide for Researchers and Drug Developers
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on navigating the critical distinctions between method verification and validation in microbiological analysis.